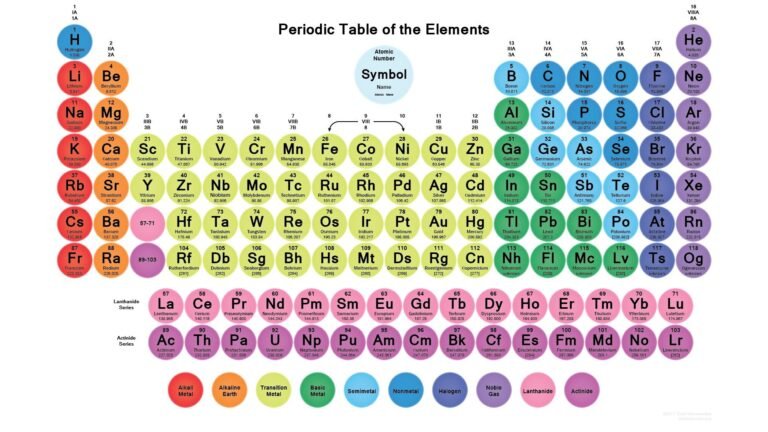
Understanding the periodic table with charges is essential for mastering chemistry. Whether you are a student preparing for exams or someone interested in chemical reactions, knowing how elements gain or lose electrons helps you predict compound formation and ionic behavior.
In this detailed, we will explore the periodic table with charges, explain ionic charges by group, discuss trends, provide memory tricks, and show how to use this knowledge in real-world chemistry problems.
What Is the Periodic Table with Charges?
The periodic table with charges refers to a version of the periodic table that highlights the common ionic charges of elements. These charges represent how many electrons an atom typically gains or loses to form a stable ion.
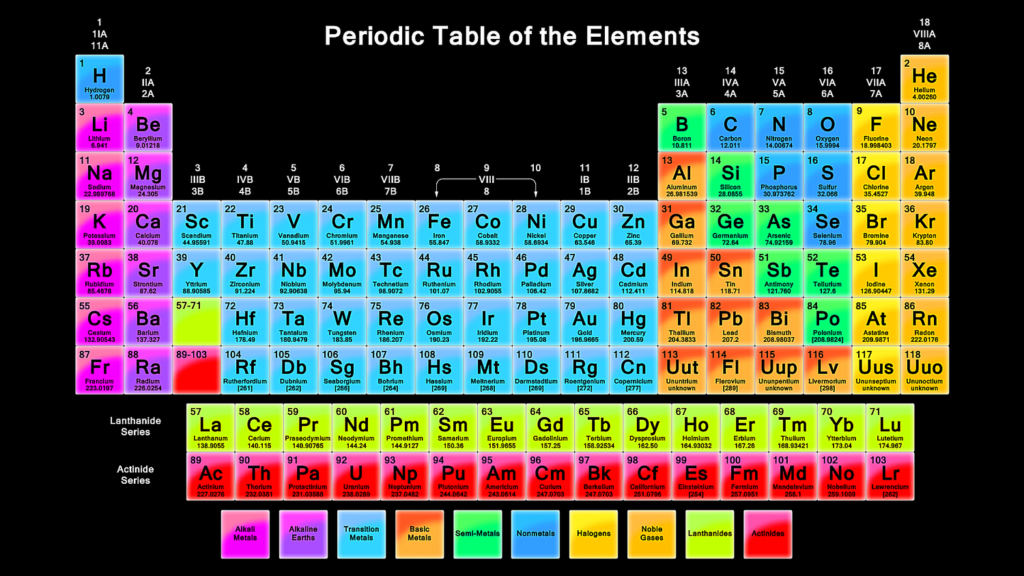
In chemistry, atoms seek stability by achieving a full outer shell of electrons. To do this, they may:
- Lose electrons (forming positive ions or cations)
- Gain electrons (forming negative ions or anions)
Understanding the periodic table with charges makes it easier to predict chemical bonding and reactions.
Why the Periodic Table with Charges Is Important
Learning the periodic table with charges helps you:
- Predict ionic compound formulas
- Balance chemical equations
- Understand reactivity trends
- Identify oxidation states
- Prepare for exams like GCSE, SAT, MCAT, and university-level chemistry
Without understanding charges, writing chemical formulas like NaCl or CaCl₂ becomes confusing.
Group Trends in the Periodic Table with Charges
One of the most helpful aspects of the periodic table with charges is that many groups follow predictable patterns.
Group 1: +1 Charge
Alkali metals typically form +1 ions.
Examples:
- Lithium (Li⁺)
- Sodium (Na⁺)
- Potassium (K⁺)
These elements lose one electron easily.
Group 2: +2 Charge
Alkaline earth metals commonly form +2 ions.
Examples:
- Magnesium (Mg²⁺)
- Calcium (Ca²⁺)
- Barium (Ba²⁺)
They lose two electrons to achieve stability.
Group 13: +3 Charge
Elements in this group often form +3 ions.
Example:
- Aluminum (Al³⁺)
Group 15: –3 Charge
Nonmetals in this group tend to gain three electrons.
Example:
- Nitrogen (N³⁻)
Group 16: –2 Charge
These elements usually gain two electrons.
Examples:
- Oxygen (O²⁻)
- Sulfur (S²⁻)
Group 17: –1 Charge
Halogens typically gain one electron.
Examples:
- Fluorine (F⁻)
- Chlorine (Cl⁻)
- Bromine (Br⁻)
Group 18: 0 Charge
Noble gases generally do not form ions because they already have a full outer shell.
Examples:
- Helium
- Neon
- Argon
The periodic table with charges clearly shows this predictable pattern across groups.
Transition Metals in the Periodic Table with Charges
Unlike main group elements, transition metals can have multiple charges.
Examples include:
- Iron (Fe²⁺ and Fe³⁺)
- Copper (Cu⁺ and Cu²⁺)
- Nickel (Ni²⁺)
Because of this variability, the periodic table with charges often lists multiple possible oxidation states for these metals.
Common Polyatomic Ions in the Periodic Table with Charges
While studying the periodic table with charges, it’s also important to know polyatomic ions:
- Nitrate (NO₃⁻)
- Sulfate (SO₄²⁻)
- Carbonate (CO₃²⁻)
- Ammonium (NH₄⁺)
These ions behave as single units in chemical reactions.
How to Use the Periodic Table with Charges to Write Formulas
Let’s understand using examples.
Example 1: Sodium Chloride
Sodium (Na⁺) has +1 charge.
Chlorine (Cl⁻) has –1 charge.
They combine in a 1:1 ratio → NaCl
Example 2: Calcium Chloride
Calcium (Ca²⁺)
Chlorine (Cl⁻)
To balance +2, we need two Cl⁻ → CaCl₂
Using the periodic table with charges, balancing becomes simple.
Periodic Table with Charges and Reactivity
Reactivity trends are closely connected to ionic charges.
- Group 1 metals are highly reactive because they easily lose one electron.
- Halogens are reactive because they gain one electron easily.
The periodic table with charges helps explain why sodium reacts violently with water and why chlorine forms salts quickly.
Memory Tricks for Periodic Table with Charges
Here’s an easy pattern:
- Group 1 → +1
- Group 2 → +2
- Group 13 → +3
- Group 15 → –3
- Group 16 → –2
- Group 17 → –1
You can remember it as a mirror effect across the periodic table
Difference Between Valency and Charge
When studying the periodic table with charges, students often confuse valency and ionic charge.
- Valency: Combining capacity of an element
- Charge: Actual electrical charge of an ion
Example: Oxygen has valency 2 and forms O²⁻ ion.
Oxidation States in the Periodic Table with Charges
Oxidation states represent electron loss or gain in reactions.
For example:
- Na in NaCl = +1
- Cl in NaCl = –1
Understanding oxidation states through the periodic table with charges is crucial in redox reactions
Periodic Table with Charges in Real Life
The concept is not just theoretical. It applies in:
- Medicine (electrolytes like Na⁺, K⁺)
- Batteries (Li⁺ in lithium-ion batteries)
- Water treatment (Ca²⁺ and Mg²⁺ hardness)
- Agriculture (nitrate and phosphate fertilizers)
Chemistry is everywhere, and the periodic table_with charges explains these processes.
Printable Periodic Table with_Charges
Students often use printable charts showing:
- Element symbols
- Atomic numbers
- Common ionic charges
- Color-coded groups
Having a visual periodic table with_charges improves learning speed.
Common Mistakes Students Make
- Assuming all metals have only one charge
- Forgetting transition metals have multiple oxidation states
- Mixing up valency and ionic charge
- Ignoring polyatomic ions
Using a clear periodic table with_charges reduces these mistakes.
Frequently Asked Questions
What is the charge of Group 1 elements?
They usually form +1 ions.
Why do halogens form –1 ions?
They need one electron to complete their outer shell.
Do noble gases form ions?
Generally, no. They already have full valence shells.
Why do transition metals have multiple charges
Because their d-orbitals allow variable electron loss.
Final Thoughts
Mastering the periodic table with_charges is one of the most important steps in learning chemistry. It simplifies ionic bonding, helps predict reactions, and makes balancing equations easier.
By understanding group trends, oxidation states, and polyatomic ions, students can confidently solve chemistry problems and perform well in exams.
The periodic table with_charges is not just a chart—it is a roadmap to understanding chemical behavior, bonding patterns, and real-world applications of science.